Dispensing (EPS)
Published on: 16th July 2013 | Updated on: 24th March 2026
Information to help pharmacy teams dispense Electronic Prescription Service (EPS) prescriptions safely, efficiently and in line with NHS requirements.
The sections below outline good practice, common issues to avoid, and practical tips to support smooth EPS workflows from receipt of a prescription through to sending the dispense message.
Endorsing and claiming information is found at the EPS submission page.
| Top tip | Summary | Advice |
| Split prescriptions | Pharmacists need to work with GPs to have a process in place to be able to identify patients who may have split prescriptions because of the clinical risks. |
Non-EPS item: There are some prescription items that cannot be transmitted electronically, for example it is currently not permitted to issue an electronic prescription for medicine outside the NHS Dictionary of medicines and devices. Patients not being able to obtain all medicines electronically has created problems. Working with GPs: The key lesson is that pharmacists need to work with GPs to have a process in place to be able to identify patients who may have split prescriptions and ensure that affected patients are aware of the risks that nomination could create. Should EPS be used for patient with a non-EPS item? Nomination may not be the best option for these patients. However if you and your local GPs wish to use EPS another option is for prescribers to include a note in the electronic message to indicate that there is also an FP10 for the patient. EPS and controlled drugs: Community Pharmacy England is continuing to call for Schedule 2 and 3 Controlled Drugs to be sent via the service. |
Related resources
Split prescription factsheet (NHS EPS team pdf):
Issuing an EPS prescription
- Prescribing staff prepare electronic prescriptions and assign them to a prescriber.
- Prescribers can:
- sign prescriptions individually or in bulk; and
- set reminders (for example, diary entries) to sign at set times during the day.
- If one prescriber is busy, practice staff can reassign a prescription to another prescriber.
- When the prescriber signs, the prescription is sent to the NHS Spine so a pharmacy can download it.
- Prescribers need the appropriate Smartcard access for their role.
Prescriber type EPS codes
- EPS messages include a code to show which prescriber type issued the prescription (for example, General Practitioner or community practitioner nurse). These codes replace the need for paper form references (such as FP10SS) in electronic messages.
- Paper forms (for example, FP10SS or FP10HNC) are still used to print paper prescriptions or EPS tokens. Tokens can be scanned by NHSBSA, but EPS pricing uses the electronic message, not the token image.
- The EPS endorsing guide (maintained by NHSBSA) lists prescriber type EPS codes (for example, “0001” indicates GP prescribing).
- The EPS specification supports three prescribing models: acute, repeat, and electronic repeat dispensing (eRD).
System safeguards
- IT system suppliers may add safeguards so prescribers do not select items they are not authorised to prescribe (for example, blocking a community practitioner nurse prescriber from issuing certain Controlled Drugs).
- IT system suppliers may also add checks on the dispensing side to prevent teams from processing items that would not be reimbursable.
- Some pharmacy IT systems visually flag the prescriber type (for example, different on‑screen colour cues). This is optional and varies by system.
Read more
- Prescriber type EPS codes factsheet (Community Pharmacy England).
Medicines Assembly
- While you are preparing an item and waiting for collection, you do not have to update the EPS status on the Spine.
Accuracy Check options
It is for the pharmacy owner to decide how the accuracy check is carried out for EPS prescriptions. Options include:
- Check against the on‑screen prescribed information:
Systems store both the prescribed item (from the EPS message) and the dispensed item (your entry if different). The screen must make it clear which view you are in. - Check against a prescription or dispensing token:
IT system suppliers must print certain data from the EPS message onto dispensing tokens without any edits. This is assessed as part of NHS England’s clinical safety checks within the Common Assurance process. - Avoid checking against dispensing labels alone:
Labels can be edited (for example, to clarify dose). Relying on labels may add risk. Some systems provide safer aids such as a separate “pick list” (for example, on a small receipt‑sized print) to minimise paper use while supporting accuracy checks.
Key point: Do not put product information into the dose instructions area.
Examples of what not to free‑type in the dose area:
- “Unlicensed”, a brand or manufacturer name, or “FS” (Free supply);
- flavour notes such as “Assorted Flavours” (use the appropriate flag instead); or
- “sugar‑free/preservative‑free” if that belongs in the product selection.
Why this matters
- Pricing: Standard EPS pricing uses the prescribed product code. Free‑typed dose text is not used for initial pricing.
- Paper prescriptions: Even with red separators, there is still a risk that free‑typed product information in the dose area may be missed during pricing.
- Safety: Patients, pharmacy teams and NHSBSA may not see or act on details buried in the dose text.
What to do if it happens
- Ask the prescriber to cancel and reissue the item so that the product information appears in the main product name (or issue a paper FP10 if EPS cannot support the item).
- EPS may not be possible if the item is not listed in:
- the NHS dm+d (it can be be checked in a dm+d browser – see our NHS dm+d page); or
- the GP system medicine list (some systems do not expose every dm+d item to prescribers, especially less common unlicensed items).
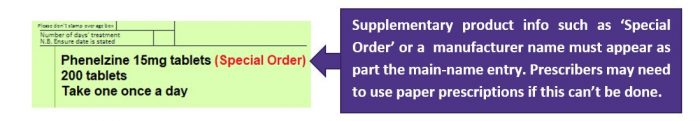
- If a prescribing system supports the correct dm+d entries (for example, “Special Order”, “Drug Tariff Special Order”, “Imported (Country)”), those terms should appear immediately after the drug name in the product line.
NHSBSA re‑checks for specials (after initial pricing)
- For specials, if the prescriber wrote “Unlicensed” in the dose area and the item was priced without that being visible, you can request an NHSBSA recheck and reprice. Include evidence (for example, a screenshot or token showing what the prescriber wrote). See: cpe.org.uk/recheck.
See also
- Avoiding EPS product info in dose area (Community Pharmacy England one‑pager) and cpe.org.uk/dosearea.
- The specials section below explains when EPS can be used for unlicensed items.

- If the GP system supports it, some unlicensed items can be issued correctly via EPS. The wording “Special Order”, “Drug Tariff Special Order” or “Imported (Country)” must appear immediately after the drug name in the product line.
- If “unlicensed” or a specials manufacturer appears in the dose area (free‑typed), ask the prescriber to cancel and reissue. NHSBSA cannot use dose‑area free text for pricing. The replacement must be a paper FP10 or an EPS prescription with the correct product wording in the main product name.
- EPS may not be possible if the item is not listed in:
- dm+d; or
- the GP system selectable medicines list (not all dm+d entries are exposed in every system).
- Prescribers should not free‑type “unlicensed special” in the dose area. See the dose area section above.
Passing clinical messages to patients
EPS Release 2 has reduced paper, so non‑routine clinical information must be passed on in other ways. Pharmacy teams and local GPs should agree a simple, reliable method to pass patient‑specific clinical messages.
About the ‘right‑hand side’
A long time back, prescribers used to use the right‑hand side of paper prescriptions for messages (for example, review dates, repeat order forms, surgery promotions). This is not always reliable for non‑routine clinical messages because:
- Many pharmacy systems do not show the right‑hand side on screen, and printing tokens is being reduced as we move towards paperless working; and
- When printed, formatting can make key points hard to spot.
Recommended approach (interim)
- Use the message to dispenser field (left‑hand side) for patient‑specific, non‑routine clinical information. Keep it clear and concise. This gives the pharmacy visibility on screen so the message can be relayed to the patient.
- Over time, prescribers have been moving more towards direct‑to‑patient digital channels (for example, Patient Facing Services, SMS messages, NHS App and NHS Notify notifications, emails etc).
Examples of non‑routine clinical information
- last repeat dispensing batch issue;
- change in dose;
- end of repeats (for example, “repeats finished—please contact surgery for review”);
- specific review arrangements (for example, book a blood test that is not routine for this patient).
Routine information (that pharmacy staff are not expected to pass on)
- patient demographics;
- prescriber details;
- standard repeat information (drug, quantity, dose; last/next issue dates; review dates when not within four weeks);
- generic promotions (for example, flu clinic);
- general health checks (for example, well woman/man, over 41s, over 75s, screening campaigns);
- surgery marketing (for example, travel clinic, physiotherapy);
- surgery opening times.
Locally agree the method
- GP and pharmacy systems can convey some information via the right‑hand side where locally agreed, but other digital routes from the GP to the patient are often better. Agree a consistent method with your local practices.
If patients ask
- Provide, on request:
- the patient’s full list of repeat medicines (as received in the EPS message);
- the review date (where more than four weeks away); and
- the number of times the medicine can be re‑ordered without a review.

- After you dispense (or while you are dispensing), send the dispense notification to the NHS Spine. You can align timing to your local workflow and system settings.
- Systems vary. For example, a message may be sent at labelling; or a barcode on the bag label can be scanned to update the status automatically. Some IT systems let you mark an item as collected (this may or may not be tied to an EPS message). Different IT systems use different status terms.
- To complete the EPS stage, mark each item as dispensed or not dispensed. You can also use the intermediate statuses with dispenser – partial and with dispenser – owing (for partial supplies or items to follow). Update the Spine after each dispensing event.
- Recommendation: Send EPS messages frequently. If an IT system failure occurs, impact is lower when messages have already reached the Spine.
- Some messages from your EPS system may also be linked to the National Patient Prescription Tracking Service (NPPTS) – which in turn can trigger messages to the patient to advise them about medicine readiness. Read more: NPPTS
- The EPS infrastructure supports recalling dispense notification and claim messages.
- Not all IT system suppliers have built this function. If yours has not, ask your IT system supplier to add it to their development roadmap.
- Remember: recalling a dispense message is separate from amending a claim.
- A post‑dated EPS prescription is held in the clinical system and released to the pharmacy on the specified date.
- Until that date:
- the pharmacy cannot view the content (so you cannot confirm details for a patient when the practice is closed); and
- you cannot prepare the item in advance.
- To avoid these problems, work with your GPs. Consider eRD as an alternative to post‑dating.
Dosage area product information
Q. I have received a prescription with supplementary product information (for example, “unlicensed” or a brand name) written in the dose instructions. Will it price correctly and is the prescription valid?
There is a high risk that dose‑area product information will be missed by the patient, the pharmacy and NHSBSA. EPS pricing is based on the prescribed product code, so free‑typed dose information is not used to calculate payment. Ask the prescriber to reissue correctly (see dose area section above and the Avoiding EPS product info in dose area one‑pager and cpe.org.uk/dosearea).
Sending dispense and claim messages
Q. I have an EPS prescription with four items, but only two are in stock. Can I dispense the two and push the prescription back so the patient gets the other two at another EPS‑enabled pharmacy?
No. You cannot split an EPS form across different pharmacies (the same rule applies to paper). If the patient needs everything urgently and will not accept an owing, return the prescription to the Spine and give a dispensing token so another EPS‑enabled pharmacy can pull it down.
Amending unclear dose instructions
Q. The dose instructions are unclear. Can I amend them so the dispensing label is correct?
Yes. Systems allow pharmacy teams to clarify or amend dose instructions on the label when needed.
Prescriber cancellation after dispensing
Q. Can a prescriber cancel a prescription that has already been dispensed?
EPS supports cancellation, but not when the status is with dispenser, with dispenser – active, dispensed or claimed. In those cases, prescribers should follow manual processes and contact the pharmacy directly. Prescribing systems show which pharmacy downloaded the item.
Returning a prescription and nomination
Q. I downloaded a nominated prescription but do not have stock. The patient now wants to change nomination to a nearby pharmacy. If I return the prescription to the Spine and change nomination, can the other pharmacy access it?
No. Once returned to the Spine, the prescription is no longer nominated. The other pharmacy will need the prescription ID to pull it down. Print a dispensing token for the patient.
Dispensing
Q. I have received an electronic prescription however when I try to dispense this, an alert is coming up in my system to indicate that there is “no dm+d mapping”. What does this mean and what should I do?
Refer to the mapping and reporting sections of cpe.org.uk/dmd
Factsheets
- EPS CDs FAQs (one‑page factsheet)
- EPS dose area: excluding product information from the dose area
- Summary of EPS‑related forms and tokens
Related webpages
For more information on this topic please email it@cpe.org.uk











